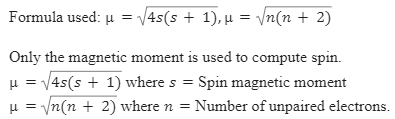The value of the spin only magnetic moment for one of the following configuration is 2.84 BM. The correct one is a)d⅝5 (in strong ligand field) b)d³ (in weak as well as

ReasonThe spin only magnetic moment of an ion is equal to sqrt {n(n+2)} where n is the number of unpaired electrons in the ion.AssertionThe spin only magnetic moment of Sc^{3+} is 1.73
What will be the theoretical value of spin only magnetic field when Fe(SCN)3 reacts with the solution containing F ions to yield a complex

![Spin only magnetic moment of the compound Hg [C0 (SCN)(4)] is Spin only magnetic moment of the compound Hg [C0 (SCN)(4)] is](https://static.doubtnut.com/ss/web/416446.webp)
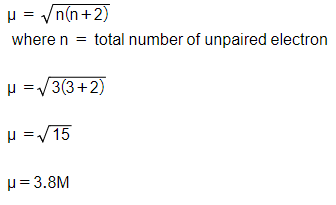



![ANSWERED] 140 The calculated spin only magnetic moment of Cr ion is 1 - Kunduz ANSWERED] 140 The calculated spin only magnetic moment of Cr ion is 1 - Kunduz](https://media.kunduz.com/media/sug-question-candidate/20201001105030189875-2167239.jpg)